Scroll to:
Bioelectronic medicine: a new frontier for autonomic nervous system disorders
https://doi.org/10.17749/2077-8333/epi.par.con.2025.231
Abstract
Bioelectronic medicine is a field of study that is constantly evolving as a result of recent advancements and improvements in bioelectronic technology, which have led to novel approaches and perspectives in disease diagnosis and therapy, particularly in the inflammatory reflex immuno-regulatory functioning and, vagus nerve stimulation (VNS). The vagus nerve, an elongated nerve in the autonomic nervous system, controls a number of physiological processes in humans, including blood pressure, breathing rate, vasomotor activity, and certain reflex movements. Recent bioelectronic research has led to clinical tests using VNS for inflammatory diseases and other conditions. By sending steady, gentle electric impulses through the vagus nerve to the brain, bioelectronic devices can activate the vagus nerve. The integration of artificial intelligence (AI) with bioelectronic medicine is transforming drug development processes. AI technology can accelerate or even eliminate many time-consuming tasks, allowing healthcare professionals to use their time more efficiently and ultimately improving healthcare outcomes. This review discusses the vagus nerve’s roles in inflammation, stimulation, and regulation in animal models, as well as its therapeutic potential in treating human inflammation. Additionally, it examines how AI-powered bioelectronic drugs are being explored for conditions such as paralysis and immune disorders, and addresses the challenges of delivering large molecules using these drugs. The article emphasizes current trends, advancements, and the promising future applications of combining AI with bioelectronic medicine.
Keywords
For citations:
Dhall M., Tushir R., Sharma P., Rani N., Singh T. Bioelectronic medicine: a new frontier for autonomic nervous system disorders. Epilepsy and paroxysmal conditions. 2025;17(3):297-307. https://doi.org/10.17749/2077-8333/epi.par.con.2025.231
INTRODUCTION / ВВЕДЕНИЕ
Steady electrical and compound signaling cells, organs, and organisms to keep up with homeostasis, which empowers different physiological outcomes to respond to the cut and contamination. Analytical treatments that synergize or affect these natural signs are expected to ease or completely resolve a clinical infirmity [1]. These days, medicines are essentially connected with compound signs, with the medications oftentimes focusing on cell receptors, chemicals, proteins, or DNA [2].
The role of electrical signals has been successfully reported in novel ways for distinct APIs' functions and to introduce an elective idea into restorative direct. In 1762, Galvani, an Italian doctor, presented the expression "bioelectricity", where he discussed how nerves and muscles communicate by sending electrical impulses through the neural system [3]. Electrical stimulation and electrophysiological recording are two methods of two-way communication with the nervous system. The first electroencephalogram, which assessed how muscles responded to an electrical cortical input and enabled the identification of somatosensory evoked potentials, led to the development of modern somatosensory maps. The motor cortex's topography has also been studied in order to quantify this reaction [4][5]. Cellular research has been done on the electrical properties of nerve cells. It has been discovered that nerve cells contain a sort of electrical energy known as membrane potential. A specific type of neuronal discharge pattern is an action potential [6][7]. Dysregulated immunological reactions that result in persistent inflammation are a distinctive component of the aetiology of rheumatoid arthritis [8].
When it comes to responding to aberrant inflammation, which is crucially important in the pathophysiology of many diseases, the use of electricity stimulates a great interest in researching the stimulation of vagus nerves and other procedures based on the inflammatory results [9][10]. In certainty, nerves not only transport essential sensory data but also messages to and from our brain, virtually every bodily module, and our organs. If the information is being communicated by electrical impulses from the nerves, it could be possible to intercept those signals and use modern sensing techniques and technology to decipher or interpret their meaning in order to extract the necessary data. The capacity to catch and decode neural impulses inside the central nervous system (CNS) has been exhibited now [11]. It has been confirmed that even in a immobilized individuals’ years after their disease, these methods can be used to infer signals in the motor region. In reality, it has been shown that paralyzed patient might control a PC cursor by just envisioning the movements of their incapacitated appendage. The developments of appendages in a immobilized patient can be re-established by decoding their decisions and once again connecting with their muscles, as expressed by the first-in-human review [12][13].
These days, neurostimulation is used to treat conditions, for example, chronic Parkinson's disease, breathing problems, gut health and sexual dysfunctions, epilepsy, Alzheimer's disease and hypertension, impulsive issues. Crohn's sickness and rrheumatoid joint pain have both been effectively treated with bioelectronic treatment already. The development of tumour necrosis factor (TNF) in the liver and the beginning of shock was repressed in rodents during endotoxemia by the excitement of the fringe vagus nerve and furthermore the tweak of aggravation and the reaction to endotoxin could be seen because of efferent vagus nerve flagging [14][15].
VAGUS NERVE FUNCTIONING / ФУНКЦИИ БЛУЖДАЮЩЕГО НЕРВА
The vagus nerve is a combination of both afferent and efferent neurons. The function of efferent neurons in the vagus nerve started in the dorsal motor nucleus (DMN) and ambiguous nucleus (NA) in the brainstem medulla oblongata, is to build up a few instinctive organs [16]. These long pre-ganglionic neurons, interconnected with post-ganglionic neurons is available in close contact or inside the innervated structures [17][18].
The significant function of the vagus nerve is to manage the pulse, GI motility, and discharge, the emission of the pancreas both endocrine and exocrine, glucose creation in the liver, and secretion. Acetylcholine (ACh) delivered by target cells, like smooth muscle cells, cardiovascular myocytes, and glandular cells, collaborates with muscarinic acetylcholine receptors (mAChRs) to carry out these roles [19].
The vagus nerve additionally contains afferent neurons, comprising ~80% of the absolute neuronal count. The pseudo-unipolar neurons' present in the jugular ganglia and nodular are missing in the focal sensory system (CNS) as displayed in Figure 1 [20].

Figure 1. Electroacupuncture at the Hegu point activates the mental mAChR, which in turn activates the efferent vagus and splenic mitigating flagging, causing somatosensory initiation. Vagus nerve and splenic nerve flagging, which intervene via splenocytes' 7nAChR, regulate the progression of severe kidney injury and ameliorate the situation [20]. Image drawn from BioRender.
mAChR – muscarinic acetylcholine receptors; AchE – acetylcholinesterase; DMN – dorsal motor nucleus; NTS – nucleus tractus solitarius; PAMPs – pathogen-associated molecular patterns; TLR4 – toll-like receptor 4; ACh – acetylcholine; NE – norepinephrine; β2AR – beta-2-adrenergic receptor; T-ChAT – T-choline acetyltransferase cells; α7nAChR – alpha-7 nicotinic acetylcholine receptor; IL-1R – interleukin-1 receptor
Рисунок 1. Электроакупунктура в точке Хэгу активирует mAChR головного мозга, которые, в свою очередь, активируют эфферентные окончания блуждающего нерва и селезеночный нерв, смягчая их возбуждение и вызывая соматосенсорную инициацию. Воздействие блуждающего нерва и селезеночного нерва, которое осуществляется через 7nAChR на спленоцитах, регулирует прогрессирование тяжелого повреждения почек и улучшает состояние пациента [20]. Рисунок сделан в программе BioRender.
mAChR (англ. muscarinic acetylcholine receptors) – мускариновые ацетилхолиновые рецепторы; AchE (англ. acetylcholinesterase) – ацетилхолинэстераза; DMN (англ. dorsal motor nucleus) – дорсальное двигательное ядро; NTS (англ. nucleus tractus solitarius) – ядро солитарного тракта; PAMPs (англ. pathogen-associated molecular patterns) – патоген-ассоциированные молекулярные паттерны; TLR4 (англ. toll-like receptor 4) – толл-подобный рецептор 4; ACh (англ. acetylcholine) – ацетилхолин; NE (англ. norepinephrine) – норадреналин; β2AR (англ. beta-2-adrenergic receptor) – бета-2-адренорецептор; T-ChAT (англ. T-choline acetyltransferase cells) – холинацетилтрансфераза Т-клеток; α7nAChR (англ. alpha-7 nicotinic acetylcholine receptor) – альфа-7 никотиновый ацетилхолиновый рецептор; IL-1R (англ. interleukin-1 receptor) – рецептор интерлейкина-1
VAGUS NERVE AND THE CHOLINERGIC ANTI-INFLAMMATORY REFLEX: MECHANISMS AND MODELS / БЛУЖДАЮЩИЙ НЕРВ И ХОЛИНЕРГИЧЕСКИЙ ПРОТИВОВОСПАЛИТЕЛЬНЫЙ РЕФЛЕКС: МЕХАНИЗМЫ И МОДЕЛИ
Regulation of inflammation by vagus nerve / Регуляция воспаления блуждающим нервом
The tissue injury and disease are primarily safeguarded with the inception of aggravation. The various receptors like toll-like receptors (TLRs), NOD-like receptors (NLRs), and other sub-atomic sensors on resistant cells and their collaboration with microorganism are significant components for commencement of the immune rsponse during inflammation [21][22]. TNF, interleukin (IL)-1, IL-6, and IL-10 are among the cytokines that are produced as a result of this interaction's stimulation of nuclear factor-κB-mediated and other intracellular signalling pathways [23][24].
The synthesis of several pro- and anti-inflammatory cytokines, enhanced leucocyte recruitment in the inflamed area, angiogenesis, and the release of resolvins and other molecules, which can result in a rapid resolution, all work together to mediate a complex and balanced inflammatory process. An extensive progression was created by the identification of the efferent vagus nerve which, has a significant impact in the neuro-safe discourse with brain control of resistant capability and irritation [25]. The role of in-rodents, regulated endotoxin lipopolysaccharide (LPS), vagotomy (a careful disturbance of the vagus nerve) is to expand the degrees of circling TNF, however a diminished degree of electrical feeling of the fringe vagus nerve in the neck. Alongside this, the focal atom set free from efferent vagus nerve axons diminishes the LPS-actuated TNF creation. This remarkable immunomodulatory action of the efferent vagus nerve was alluded to as a cholinergic mitigating pathway. It was likewise found that the receptor responsible for changing over cholinergic calming outpouring from the vagus nerve into a lessening in the age of proinflammatory cytokines is the nicotinic acetylcholine receptor (7nAChR), which is situated on macrophages and other safe cells [12][26].
The communication of the vagus nerve with the splenic nerve manages the aggravation reflex. It was fundamentally shown by addressing the concealment of splenic TNF and serum vagus nerve stimulation (VNS) in murine endotoxemia, which was repealed by splenic nerve crosscut. VNS additionally accelerates the arrival of ACh in the spleen. A small part of lymphocytes that have choline acetyltransferase (Talk), a pivotal protein in ACh fabricate, are a huge cell wellspring of this ACh. These lymphocytes with choline acetyltransferase (T-Visit) cells work with catecholaminergic actuation of ACh discharge heavily influenced by the vagus nerve as well as communicating-adrenergic receptors. As displayed in Figure 2, ACh likewise reacts with the alpha-7nAChR.
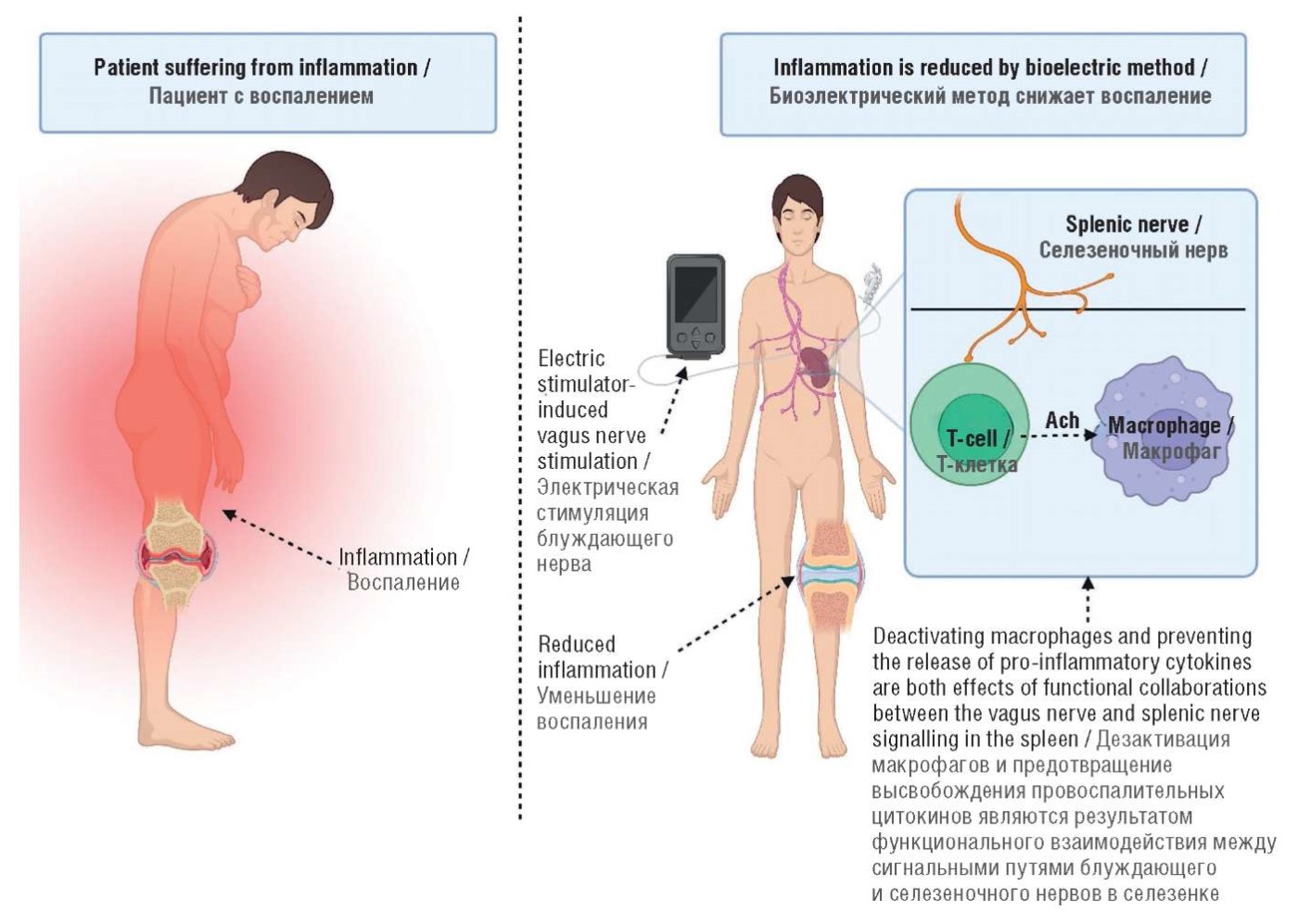
Figure 2. A bioelectronic technique that targets the vagus nerves to reduce inflammation is shown schematically. Inflammation is decreased by acetylcholine, which is produced by splenic T-cells through an interaction with the splenic nerve and the vagus nerve. Image drawn from BioRender.
ACh – acetylcholine
Рисунок 2. Схема воздействия биоэлектронной технологии на блуждающий нерв для снижения воспаления. Воспаление уменьшается благодаря ацетилхолину, который вырабатывается Т-клетками селезенки при взаимодействии с селезеночным и блуждающим нервами. Рисунок сделан в программе BioRender.
ACh (англ. acetylcholine) – ацетилхолин
Regulating the inflammatory reflex and inflammation via brain signalling / Регулирование воспалительного рефлекса и воспаления посредством сигналов мозга
Guanylhydrazone’s (the vagus nerve's mitigating action) action is completely supported by the research involving the studies on anti-inflammatory chemical known as CNI-1493. The vagus nerves were found to be a significant brain-to-periphery transporter of CNI-1493 calming activities. CNI-1493 given in the cerebrum was found to diminish fringe cytokine levels. New brain restorative targets would be uncovered by an intensive planning of the mind networks that control the incendiary reflex and the vagus nerve's calming capability. In the future, bioelectronic medicine may be able to use these techniques to regulate brain networks to treat inflammatory diseases. Utilizing AI in obtaining the required role of inflammatory actions is achieving the in-sights in the field of advanced pharmaceutical technologies [13][27][28].
Inflammatory VNS: animal models / VNS при воспалении: животные модели
Numerous studies employing VNS have revealed mechanistic information on the vagus nerve's anti-inflammatory effect and suggested new ways to treat inflammation. Concentrating on the cerebrum components in this field presents an entrancing region for impending exploration. Stimulation of vagus nerve has a potential to operate following functions: uncontrolled bleeding, murine endotoxemia, postoperative ileus, kidney ischemia-reperfusion injury, sepsis, haemorrhagic shock, and different problems. Aggravation and other obsessive parts of collagen-prompted joint pain in rodents are enormously diminished by once-everyday VNS managed through an embedded sleeve cathode for seven days [29].
The seriousness of inflammatory bowel disease (IBD) (colitis) and colonic irritation are enormously decreased in rodents when a tantamount strategy – three hours of VNS excitement each day utilizing an embedded gadget – is utilized. Endotoxemia in mice resulted in a feeling of the cervical vagus nerve with a percutaneous needle terminal embedded under ultrasound direction, which fundamentally decreases fringe and CNS irritation (neuroinflammation). Aggravation control strategies utilizing painless VNS have additionally been examined. One of these procedures is vagus nerve stimulation in the auricular branch. The control of dying (discharge), body weight, and its suggestions for immunoregulation and incendiary administration have all been utilized to show the convenience of VNS [30][31].
VNS in human inflammatory diseases / VNS при воспалительных заболеваниях человека
Conditions including rheumatoid IBD, joint pain, and weight aggravation exist together with diminished vagus not entirely set in stone by pulse fluctuation examination. Results from bioelectronic VNS clinical examinations in patients with rheumatoid joint pain and IBD (Crohn's illness) have started to approve the translational relevance of preclinical discoveries. Crohn's sickness is a persistent provocative infection with restricted, costly, and related with broad unfavourable impact therapy choices [32][33]. In 5 out of 7 patients with dynamic sickness, VNS utilizing an embedded gadget decisively diminished the seriousness of the condition as proven by a diminished illness action score and better endoscopic outcomes [27]. Patients with this condition who got bioelectronic VNS (up to 4 times each day) utilizing an embedded gadget experienced huge illness alleviation, as seen by perceptibly better infection scores, for as long as 84 days [34]. Two patient partners – 7 patients in an early sickness stage showed no reaction to earlier methotrexate therapy and 10 people at a later illness stage who bombed treatments with a few biologics – answer well to this VNS treatment. Following VNS, clinical improvement in these rheumatoid joint pain patients happens simultaneously with drops in TNF and other cytokine levels [35].
BIOELECTRONICS IN IMMUNITY / БИОЭЛЕКТРОНИКА В ИММУННОЙ СИСТЕМЕ
Organization of nervous system / Организация нервной системы
Nervous system consists of two parts, i.e. peripheral and CNS. The peripheral system has physical nerves that starts from the CNS, innervate skeletal muscles, and give voluntary control of actions. The autonomic nervous system has sympathetic, parasympathetic, and intestinal parts [36]. The neurotransmitter signals are transferred through synapses between postganglionic strands and sensory neurons arranged in the spinal cord innervate veins, lymphoid tissue and organs, bone marrow, the lungs and aviation routes, gastrointestinal tract, liver, kidneys, and other instinctive organs. While ganglionic synaptic neurotransmission is cholinergic, postganglionic neurons discharge norepinephrine and, less significantly, different catecholamines (like adrenaline and dopamine), as well as neuropeptide Y [28].
Regulation of neuronal reflex / Регуляция нейронального рефлекса
Neuroscientists in the mid-20th century worked for a very long time to thoroughly plan reflexes for regulating functional homeostasis in individuals. The reflex was named after Harvey Cushing, the pioneer behind current neurosurgery. He found it by setting an inflatable in a canine's head and looking for changes in pulse, pulse, and breath rate that happened subsequently [37]. He isolated the vagus nerves to more readily comprehend the mind networks controlling these physiological responses, and he found that he could isolate the circulatory strain and pulse reactions that happened because of ascends in intracranial tension.
By the middle of the 20th century, these and other related tests led to the identifiable evidence of several reflexes, such as the vagus nerve and other nerves, as tools to regulate the homeostasis of almost every organ in the body, but not the safe framework. An example of a reflex that is both very simple and extremely complex, including several different types of neurones and central nervous system cooperation, is the axon reflex. Natural alterations are detected by tangible neurones, which then trigger an axon reflex response [24][38].
Signaling between the nervous and immune systems / Сигналы между нервной и иммунной системами
Niels Kaj Jerne quickly pointed towards the similarities for the anxious and invulnerable frameworks, as well as acknowledgment parts and the capacity to learn and shape recollections. One important source of indicators that affect the insusceptible framework's capacity and indicators from it is the sensory system [39]. These associations are worked with by physical proximity and sharing sub-atomic sources of correspondence, including receptors and flagging particles. The skin and instinctive organs are innervated by afferent and efferent nerves, which are decisively positioned to screen harm and contamination destinations. Tactile neurons have been found to communicate particles that were beforehand just connected with resistant control, like example recognition receptors (like TLRs) and receptors for TNF, IL-1, and different cytokines [40][41]. Also, it had revealed that resistant cells express receptors that have generally been connected to fringe nerve guideline and brain transmission in the focal sensory system.
Sensory neurons and immune challenges / Сенсорные нейроны и иммунные проблемы
The body's all organs and tissues are innervated by afferent neurons, which go about as a vital channel for the CNS to get data about fringe changes in immunological homeostasis. Microbes and synthetically resistant substances animate tangible neurons that have focal associations with the cell bodies and spinal cord in the dorsal root ganglia [42][43]. These neurons along-with hand-off neurons and spinal interneurons transfer the reflexes to the mind inside the spinal cord. The essential subset of these neurons, known as nociceptors, is specialists in communicating various sorts of torment, which is likewise a pivotal part of irritation [13].
On tactile neurons, the statement of different voltage-gated sodium channels as well as transient receptor potential particle channels intervenes in depolarization and explicit warm, mechanical, and substance aversions to harmful boosts [13]. Through these receptors, cytokines like TNF, IL-1, IL-6, IL-17, prostaglandins, and different synthetic substances produced by adjacent invulnerable cells like neutrophils, pole cells, and macrophages associate with tangible neurons during disease, sensitivities, and tissue injury. At the point when there is a bacterial or viral contamination, cell harm, or an allergic response, vagal sensory neurons become dynamic [44][45].
TNF, IL-1, prostaglandins, serotonin, and different synthetic compounds created by resistant cells, like neutrophils, macrophages, and eosinophils, play a part in this enactment. When regulated to mice and rodents, lipopolysaccharide (LPS), IL-1 and TNF and contaminations such as Campylobacter jejuni, vagus nerve afferent transmission is activated. This flagging might be followed by the NTS to different brainstem and forebrain areas. These outcomes recommend that changed fringe immunological signs might influence afferent vagus nerve action in an unexpected way [46][47]. The various bioelectronic medicine approaches has been tabulated in Table 1.
Table 1. Summary for bioelectronic medicine approaches
Таблица 1. Краткое описание подходов биоэлектронной медицины
|
Condition / Состояние |
Clinical result / Клииический результат |
Mechanism / Механизм |
Reference / Источник |
|
Rheumatoid arthritis / Ревматоидный артрит |
Decrease in severity of the disease using VNS, as determined by standardized clinical composite scores / Уменьшение тяжести заболевания при использовании VNS по оценке стандартизированных клинических комплексных показателей |
The production of TNF and other pro-inflammatory cytokines are inhibited by the activation of neural circuitvia macrophages / Выработка TNF и других провоспалительных цитокинов подавляется при активации нейронных сетей через макрофаги |
[48] |
|
Crohn’s disease / Болезнь Крона |
Among 7 participants 5 are evolved towards biological, clinical and endoscopic remission with a restored vagal tone after VNS / Из 7 участников у 5 наблюдается развитие биологической, клинической и эндоскопической ремиссии с восстановлением тонуса блуждающего нерва после VNS |
– Afferents neurons activate the hypothalamic-pituitary-adrenal axis / Афферентные нейроны активируют гипоталамо-гипофизарно-надпочечниковую ось – Efferent neurons activate the cholinergic anti-inflammatory pathway / Эфферентные нейроны активируют холинергический противовоспалительный путь |
[49] |
|
Paralysis / Паралич |
C5–C6 to C7–T1 level motor impairment level improved allowing participants to hold and manipulate diverse size objects with different grips / Снижение двигательных нарушений на уровне от C5–C6 до C7–T1 позволило испытуемым удерживать и манипулировать предметами разных размеров с помощью разных захватов |
Activation of muscles by neuromuscular stimulation is regulated by decoded intra-cortically recorded signals from the motor cortex / Активация мышц посредством нервно-мышечной стимуляции регулируется декодированными интракортикальными сигналами от двигательной коры |
[50] |
Note. VNS – vagus nerve stimulation; TNF – tumor necrosis factor.
Примечание. VNS (англ. vagus nerve stimulation) – стимуляция блуждающего нерва; TNF (англ. tumor necrosis factor) – фактор некроза опухоли.
Decoding hidden messages / Декодирование скрытых сообщений
Brain deciphering includes the area of signal handling and is a part of the search of man-made reasoning called AI to get familiar with the 'language of the sensory system'. This assists in diagnosing conditions such as Alzheimer’s, Parkinson’s, schizophrenia, and fanatical enthusiastic issue significantly quicker than conventional strategies [51]. Essential examinations established the groundwork for the resulting brain unravelling explores associated with concentrating on the strategies to encode the data connected with tangible and engine capabilities. Further advancements in brain unravelling, and cerebrum PC interface (Brain-Computer Interface, BCI) innovation, prompted the examinations including human members existing with amyotrophic lateral sclerosis, stroke and also spinal rope injury [52–55].
In these underlying examinations, BCI and brain translating technology effectively permitted members to control the devices like PCs, mechanical arms, and an electric wheelchair through their solely with their eyes. Brain interpreting has basically been a field that has zeroed in on unravelling brain action connected with tangible, engine or even illness states inside the mind. In any case, the brain interpretation techniques that had been produced for the mind can be adjusted to the remainder of the sensory system later on. The single vagus nerve has roughly 100,000 filaments of which around 80% strands are afferent (tactile) filaments, conveying data from different organs and portions of the body to the cerebrum [56–59].
Treatment of paralysis via technology / Лечение паралича с помощью новых технологий
Previous BCI innovation permitted paralyzed patients to regulate PC cursors and wheel-chairs made ready for rebuilding development in their appendages. Early non-human examinations were briefly on incapacitated arm muscles which were initiated heavily influenced by a BCI. Later on, brain side-step’s innovation was created and shown in a first-barbaric review including a 23-year-old male quadriplegic member. A small terminal exhibit 96 cathodes with (4×4 mm across) was embedded in the brain cortex and using the brain interpretation technique; the review member had the option to rotate his paralyzed hand under the control of volitional. The member had the option to successfully recapture volitional finger and wrist developments by his own contemplations and to begin and shut down those developments when needed.
DEVICES AND MATERIALS FOR UNCOVERING NEW BIOLOGY / УСТРОЙСТВА И МАТЕРИАЛЫ ДЛЯ РАСКРЫТИЯ НОВОЙ БИОЛОГИИ
Due to improvements in micro-fabrication and novel materials, the bioelectronic interface has now been reduced from large scale connections capable of triggering whole muscles to gadgets that can link with specific cells and membrane proteins [60]. Due to their vast variety of extent scales, these bioelectronic strategies are the finest technique to study and activate bioelectric circuits.
Biosensors / Биосенсоры
Biological markers, electric fields, and ionic concentrations can all be detected using bioelectronic sensors. Extensive categories of bioelectronic sensors were covered in past evaluations. Cell-sensors, or collections of cells, are the principal focus of both extracellular detection and intracellular recordings [61][62].
Extracellular neural activities / Внеклеточная нейронная активность
Biosensors like transistor-type provide signal magnification and extensive level multiplexing. A well-liked construction method for transistor-based biosensors (organic electrochemical transistor – OECT) is the organic electrochemical transistor. Poly (3, 4-ethylene di-oxythiophene): poly (styrene sulfonate) (PEDOT:PSS) was mixed with certain enzymes or antibodies to make a conventional OECT in order to identify compounds of interest [60]. Due to the conducting channel's capacity to carry both electrons and ions, OECT has an intrinsic sensitivity to ionic classes in solution. In light of this, OECT behaves like a transducer between the two kinds of charge carriers. The health of the cells and how the barrier tissue responds to external substances may both be assessed using this transduction technique. three-dimensional tissue [60][63].
Intracellular recording / Внутриклеточная запись
The novel methodologies of microfabrication and nanomaterials permit the bioelectronic devices to mimic the receptor-mediated response of solitary cells to record membrane potential (voltage across the cell membrane – Vmem) [64]. These signals are based on the advanced procedure of fix bracing. Most bioelectronic methods are spun around estimating spatiotemporal planning of Vmem of mechanized sensitive cells, like cardiomyocytes and neurons. Silicon nanowires with high perspective proportions and nanoscale distances across can interface the intracellular space and cross the cell layer with practically no mischief to the cell [65][66]. Vmem denotes the electrical potential difference between the outside and inside of a cell, typically produced by ion gradients (like Na+, K+, Cl–, Ca2+).
In neurons, Vmem is critical for action potential generation and signal transmission. In non-excitable cells, Vmem also regulates processes such as cell proliferation, differentiation, migration, and tissue regeneration.
By changing the doping levels, a nanoscale field impact semiconductor might be made on a solitary nanowire due to the spatially controlled electrical signals of these nanowires. These crimped nanowires are framed to join to the tip of a nuclear power magnifying lens test and be embedded into the specific cell. Using silicon nanowire, enhances the potential cardiomyocyte activity inside the cell as compared to outside [28][67][68].
Bio-actuators / Биоактуаторы
Galvani's initial work and contemporary clinical devices like pacemakers and brain inserts exhibit how bioelectric correspondence to cells arises over an electronic drive at the terminals that influences the activity capability of mechanized volatile cells [69]. Here, we'll focus on bioelectronic device that can convey biophysical and synthetic signals straightforwardly to target specific cell processes.
The electrophoretic exchange of particles and minuscule atoms, like synapses, to change cell action is one of these signs. To move particles and little charged particles from a supply well into mass arrangement, natural electronic particle siphons made of PEDOT:PSS should undergo for ionically conductive poly-particle trade layer as well as with PEDOT:PSS anode. This makes different charged species be siphoned, including monovalent cations, gamma-aminobutyric acid, and acetylcholine. These natural particle siphons can ship various particles as well as charged atoms, yet the adequacy of their conveyance diminishes as the size of the particles gets bigger. It is possible to transport larger particles due to the directed penetrability of the dendrimer's ionic polymer, which permits increased particle transit [70].
ROLE OF AI IN BIOELECTRONIC MEDICINE / РОЛЬ ИИ В БИОЭЛЕКТРОННОЙ МЕДИЦИНЕ
The emotionally supportive network should focus on a number of other extremely delicate issues, albeit an early determination would be extremely helpful, yet in addition on various other exceptionally delicate issues, for instance, a superior emergency, to comprehend which patients should be visible first. As a matter of fact, an early discovery of hostile instances, or in any event, featuring a few heinous acts that could prompt clinical mistakes, would be of extraordinary help.
The utilization of man-made perceptive in medication traces all the way back to the 1960s. A few endeavours to help clinical finding utilizing man-made reasoning were made then, at that point, for example, to recognize hazardous microscopic organisms and propose suitable treatment. Curiously, that framework had the option to propose great treatment with preferred execution over irresistible infection specialists. Following are the some key-points indicating role of AI in medicine:
– signal decoding – AI helps analyze and interpret complex neural signals;
– pattern recognition – AI identifies disease-specific electrical activity in the nervous system;
– therapy optimization – AI adjusts stimulation parameters in real time for better outcomes;
– personalized medicine – AI predicts patient response and enables tailored treatments;
– closed-loop systems – AI supports adaptive devices that respond instantly to physiological changes.
Pharmaceutical companies are now using AI to find more effective drug candidates and filter out those that could be harmful to humans before they go very far in the pipeline. Cutting out the weak drug candidates early on with AI saves millions of dollars and years of development [71].
RECENT TRENDS AND DEVELOPMENTS / ПОСЛЕДНИЕ ТЕНДЕНЦИИ И РАЗРАБОТКИ
VNS holds the capability of giving productive method for treating an broad range of humanoid conditions, including IBD, joint pain (rheumatoid), ileus, abandoned dying, and the cutting-edge contagions – obesity and the firmly linked metabolic condition and type 2 diabetes. Translational improvements in bioelectronic medication followed by on-going preclinical and clinical examinations that are revealing novel insights into the capability of the vagus nerve in the control of irritation and restorative adequacy of the VNS, as explained previously.
Beyond vagus nerve modulation, bioelectronic medicine research also examines the regulatory mechanisms of other peripheral and central nervous system circuits and how to modify them therapeutically. Potential uses in a variety of illnesses, such as cancer, neurological disorders, diabetes, paralysis, cardiovascular diseases, and vision impairment, are being investigated in ongoing clinical studies. This field seeks to address disease progression and restore physiological equilibrium through precise modulation of bioelectrical signals. Furthermore, expanding bioelectronic interventions outside traditional neurological regulation presents both opportunities and concerns, as highlighted in contemporary discourse.
The essential focal point of bioelectronic medication is still brain mediation, albeit this field has drawn a lot of interest and consideration from both the general population and private areas, with GlaxoSmithKline (GSK) starting to lead the pack and putting more than $50 million USD in bioelectronic gadgets for therapeutic bids1. Run-of-the-mill electroceuticals connect with nerves and have gone through broad testing because of the great sensitivity of brain tissues and powerlessness to electrical excitement. It has various helpful purposes from vagus nerve triggers that treat inflammation and immune system diseases to instances of vision restoration with the cochlear implant.
Nonetheless, bioelectronics has been to a great extent ignored in the field of bioelectronic medication, notwithstanding being available in all cell types as electric fields, ionic flows, redox responses, and expected contrasts. Endogenous non-brain bioelectric motivations can make cells open and close ionic channels and actuate processes for quality record. These pathways can intercede the correspondence between safe cells and tissues and are participated in various cell processes, including movement, separation, and division [51]. Medicines now accessible have harmfulness and aftereffects, including methotrexate and biologics including against TNF, hostile to IL-6 receptor, against CD20 antibodies, and white blood cell co-excitement inhibitor. Also, certain individuals don't profit from these treatments. The vagus nerve was invigorated for as long as 84 days by a bioelectronic embed in two gatherings of 17 patients, one of whom had bombed methotrexate treatment or treatment including at least two biologics, and the other of whom was in the early stages of the sickness and not answering methotrexate. The assembling of TNF is likewise diminished by this bioelectronic neuromodulation.
FUTURE PERSPECTIVES / ПЕРСПЕКТИВЫ
The majority of bioelectronic medical devices, such as deep brain stimulators, have a tendency to be quite intrusive. The nanoscale characteristics along with soft biocompatible materials reduce the invasiveness of the bioelectronic devices. Prof. Lieber's team at the University of Harvard confirmed the reduced invasiveness in bioelectronics injectables with some experiments like improving the flexible wearable electronics and tattoos by applying them directly to the patient’s skin, resulting in improved diagnostic capabilities. Future devices will deliver highly targeted stimulation to specific branches of the ANS, reducing side effects and improving therapeutic efficiency. Integration of biosensors with AI will allow real-time monitoring of physiological signals and adaptive control of stimulation. Devices will be tailored to individual patient profiles, enabling customized treatment plans for disorders such as hypertension, arrhythmias, and inflammatory diseases for personalized therapy.
CONCLUSION / ЗАКЛЮЧЕНИЕ
Focusing on the fiery reflex utilizing VNS can be a proficient new way to control variant aggravation in numerous illnesses. Worked on comprehension of the sensory system administrative capabilities and their changes in preclinical sickness models will keep on giving vital unthinking bits of knowledge to additional clinical preliminaries. Constant improvement of assembling methods and mechanical headways are driving a quick development in the bioelectronic medication field. In future, there could be critical headways in the field of bioelectronic medication, which coordinates and deciphers information about atomic standards of mind control and sickness causation into novel therapies for human illness based on neuromodulation of discrete neurocircuitries.
1. https://www.gsk.com/en-gb/media/press-releases/gsk-launches-50-million-venture-capital-fund-to-invest-in-pioneering-bioelectronic-medicines-and-technologies.
References
1. Löffler S., Melican K., Nilsson K.P.R., Richter-Dahlfors A. Organic bioelectronics in medicine. J Intern Med. 2017; 282 (1): 24–36. https://doi.org/10.1111/joim.12595.
2. Zhang J., Wang X., Vikash V., et al. ROS and ROS-mediated cellular signaling. Oxid Med Cell Longev. 2016: 2016: 4350965. https://doi.org/10.1155/2016/4350965.
3. Drews J. Drug discovery: a historical perspective. Science. 2000; 287 (5460): 1960–4. https://doi.org/10.1126/science.287.5460.1960.
4. López-Muñoz F., Alamo C. Monoaminergic neurotransmission: the history of the discovery of antidepressants from 1950s until today. Curr Pharm Des. 2009; 15 (14): 1563–86. https://doi.org/10.2174/138161209788168001.
5. Fritsch G., Hitzig E. Electric excitability of the cerebrum (Über die elektrische Erregbarkeit des Grosshirns). Epilepsy Behav. 2009; 15 (2): 123–30. https://doi.org/10.1016/j.yebeh.2009.03.001.
6. Penfield W., Boldrey E. Somatic motor and sensory representation in the cerebral cortex of man as studied by electrical stimulation. Brain. 1937; 60 (4): 389–443.
7. Dawson G.D. Investigations on a patient subject to myoclonic seizures after sensory stimulation. J Neurol Neurosurg Psychiatry. 1947; 10 (4): 141–62. https://doi.org/10.1136/jnnp.10.4.141.
8. Haas L. Hans Berger (1873–1941), Richard Caton (1842–1926), and electroencephalography. J Neurol Neurosurg Psychiatry. 2003; 74 (1): 9. https://doi.org/10.1136/jnnp.74.1.9.
9. Pearce J.M.S. Emil Heinrich Du Bois-Reymond (1818–96). J Neurol Neurosurg Psychiatry. 2001; 71 (5): 620. https://doi.org/10.1136/jnnp.71.5.620.
10. Hodgkin A.L., Huxley A.F. A quantitative description of membrane current and its application to conduction and excitation in nerve. J Physiol. 1952; 117 (4): 500–44. https://doi.org/10.1113/jphysiol.1952.sp004764.
11. Pavlov V.A., Chavan S.S., Tracey K.J. Bioelectronic medicine: from preclinical studies on the inflammatory reflex to new approaches in disease diagnosis and treatment. Cold Spring Harb Perspect Med. 2020; 10 (3): a034140. https://doi.org/10.1101/cshperspect.a034140.
12. Koopman F.A., Chavan S.S., Miljko S., et al. Vagus nerve stimulation inhibits cytokine production and attenuates disease severity in rheumatoid arthritis. Proc Natl Acad Sci USA. 2016; 113 (29): 8284–9. https://doi.org/10.1073/pnas.1605635113.
13. Bonaz B., Sinniger V., Hoffmann D., et al. Chronic vagus nerve stimulation in Crohn's disease: a 6-month follow-up pilot study. Neurogastroenterol Motil. 2016; 28 (6): 948–53. https://doi.org/10.1111/nmo.12792.
14. Bouton C. Cracking the neural code, treating paralysis and the future of bioelectronic medicine. J Intern Med. 2017; 282 (1): 37–45. https://doi.org/10.1111/joim.12610.
15. Hochberg L.R., Serruya M.D., Friehs G.M., et al. Neuronal ensemble control of prosthetic devices by a human with tetraplegia. Nature. 2006; 442 (7099): 164–71. https://doi.org/10.1038/nature04970.
16. Bouton C.E., Shaikhouni A., Annetta N.V., et al. Restoring cortical control of functional movement in a human with quadriplegia. Nature. 2016; 533 (7602): 247–50. https://doi.org/10.1038/nature17435.
17. Klein E. Models of the patient-machine-clinician relationship in closed-loop machine neuromodulation. In: van Rysewyk S., Pontier M. (Eds) Machine medical ethics. Intelligent systems, control and automation: science and engineering, vol 74. Springer; 2014: 273–90. https://doi.org/10.1007/978-3-319-08108-3_17.
18. Tepper S.J., Rezai A., Narouze S., et al. Acute treatment of intractable migraine with sphenopalatine ganglion electrical stimulation. Headache. 2009; 49 (7): 983–9. https://doi.org/10.1111/j.1526-4610.2009.01451.x.
19. Deuschl G., Schade-Brittinger C., Krack P., et al. A randomized trial of deep-brain stimulation for Parkinson's disease. N Engl J Med. 2006; 355 (9): 896–908. https://doi.org/10.1056/NEJMoa060281.
20. Zhang L., Wu Z., Zhou J., et al. Electroacupuncture ameliorates acute pancreatitis: a role for the vagus nerve-mediated cholinergic anti-inflammatory pathway. Front Mol Biosci. 2021: 8: 647647. https://doi.org/10.3389/fmolb.2021.647647.
21. Sun F.T., Morrell M.J., Wharen R.E. Jr. Responsive cortical stimulation for the treatment of epilepsy. Neurotherapeutics. 2008; 5 (1): 68–74. https://doi.org/10.1016/j.nurt.2007.10.069.
22. Greenberg B.D., Malone D.A., Friehs G.M., et al. Three-year outcomes in deep brain stimulation for highly resistant obsessive-compulsive disorder. Neuropsychopharmacology. 2006; 31 (11): 2384–93. https://doi.org/10.1038/sj.npp.1301165.
23. Medzhitov R. Origin and physiological roles of inflammation. Nature. 2008; 454 (7203): 428–35. https://doi.org/10.1038/nature07201.
24. Chavan S.S., Tracey K.J. Essential neuroscience in immunology. J Immunol. 2017; 198 (9): 3389–97. https://doi.org/10.4049/jimmunol.1601613.
25. Laxton A.W., Tang-Wai D.F., McAndrews M.P., et al. A phase I trial of deep brain stimulation of memory circuits in Alzheimer's disease. Ann Neurol. 2010; 68 (4): 521–34. https://doi.org/10.1002/ana.22089.
26. Tracey K.J. The inflammatory reflex. Nature. 2002; 420 (6917): 853–9. https://doi.org/10.1038/nature01321.
27. Borovikova L.V., Ivanova S., Zhang M., et al. Vagus nerve stimulation attenuates the systemic inflammatory response to endotoxin. Nature. 2000; 405 (6785): 458–62. https://doi.org/10.1038/35013070.
28. Rosas-Ballina M., Olofsson P.S., Ochani M., et al. Acetylcholine-synthesizing T cells relay neural signals in a vagus nerve circuit. Science. 2011; 334 (6052): 98–101. https://doi.org/10.1126/science.1209985.
29. Berthoud H.R. The vagus nerve, food intake and obesity. Regul Pept. 2008; 149 (1-3): 15–25. https://doi.org/10.1016/j.regpep.2007.08.024.
30. Masi E.B., Valdés-Ferrer S.I., Steinberg B.E. The vagus neurometabolic interface and clinical disease. Int J Obes. 2018; 42 (6): 1101–11. https://doi.org/10.1038/s41366-018-0086-1.
31. Berthoud H.R., Neuhuber W.L. Functional and chemical anatomy of the afferent vagal system. Auton Neurosci. 2000; 85 (1-3): 1–17. https://doi.org/10.1016/S1566-0702(00)00215-0.
32. Akira S., Uematsu S., Takeuchi O. Pathogen recognition and innate immunity. Cell. 2006; 124 (4): 783–801. https://doi.org/10.1016/j.cell.2006.02.015.
33. Andersson U., Tracey K.J. Reflex principles of immunological homeostasis. Annu Rev Immunol. 2012; 30: 313–35. https://doi.org/10.1146/annurev-immunol-020711-075015.
34. Wang H., Yu M., Ochani M., et al. Nicotinic acetylcholine receptor α7 subunit is an essential regulator of inflammation. Nature. 2003; 421 (6921): 384–8. https://doi.org/10.1038/nature01339.
35. Tan C., Yan Q., Ma Y., et al. Recognizing the role of the vagus nerve in depression from microbiota-gut brain axis. Front Neurol. 2022; 13: 1015175. https://doi.org/10.3389/fneur.2022.1015175.
36. Berthoud H.R., Powley T.L. Interaction between parasympathetic and sympathetic nerves in prevertebral ganglia: morphological evidence for vagal efferent innervation of ganglion cells in the rat. Microsc Res Tech. 1996; 35 (1): 80–6. https://doi.org/10.1002/(SICI)1097-0029(19960901)35:1<80::AID-JEMT7>3.0.CO;2-W.
37. Bernik T.R., Friedman S.G., Ochani M., et al. Pharmacological stimulation of the cholinergic antiinflammatory pathway. J Exp Med. 2002; 195 (6): 781–8. https://doi.org/10.1084/jem.20011714.
38. Metz C.N., Pavlov V.A. Vagus nerve cholinergic circuitry to the liver and the gastrointestinal tract in the neuroimmune communicatome. Am J Physiol Gastrointest Liver Physiol. 2018; 315 (5): G651–8. https://doi.org/10.1152/ajpgi.00195.2018.
39. Levine Y.A., Koopman F.A., Faltys M., et al. Neurostimulation of the cholinergic anti-inflammatory pathway ameliorates disease in rat collagen-induced arthritis. PLoS One. 2014; 9 (8): e104530. https://doi.org/10.1371/journal.pone.0104530.
40. Meregnani J., Clarençon D., Vivier M., et al. Anti-inflammatory effect of vagus nerve stimulation in a rat model of inflammatory bowel disease. Auton Neurosci. 2011; 160 (1-2): 82–9. https://doi.org/10.1016/j.autneu.2010.10.007.
41. Huffman W.J., Subramaniyan S., Rodriguiz R.M., et al. Modulation of neuroinflammation and memory dysfunction using percutaneous vagus nerve stimulation in mice. Brain Stimul. 2019; 12 (1): 19–29. https://doi.org/10.1016/j.brs.2018.10.005.
42. Czura C.J., Schultz A., Kaipel M., et al. Vagus nerve stimulation regulates hemostasis in swine. Shock. 2010; 33 (6): 608–13. https://doi.org/10.1097/SHK.0b013e3181cc0183.
43. Lindgren S., Stewenius J., Sjölund K., et al. Autonomic vagal nerve dysfunction in patients with ulcerative colitis. Scand J Gastroenterol. 1993; 28 (7): 638–42. https://doi.org/10.3109/00365529309096103.
44. Pavlov V.A., Chavan S.S., Tracey K.J. Molecular and functional neuroscience in immunity. Annu Rev Immunol. 2018; 36: 783–812. https://doi.org/10.1146/annurev-immunol-042617-053158.
45. Elenkov I.J., Wilder R.L., Chrousos G.P., Vizi E.S. The sympathetic nerve – an integrative interface between two supersystems: the brain and the immune system. Pharmacol Rev. 2000; 52 (4): 595–638.
46. Jänig W. Sympathetic nervous system and inflammation: a conceptual view. Auton Neurosci. 2014; 182: 4–14. https://doi.org/10.1016/j.autneu.2014.01.004.
47. Jänig W., Keast J.R., McLachlan E.M., et al. Renaming all spinal autonomic outflows as sympathetic is a mistake. Auton Neurosci. 2017; 206: 60–2. https://doi.org/10.1016/j.autneu.2017.04.003.
48. Schaible H.G. Nociceptive neurons detect cytokines in arthritis. Arthritis Res Ther. 2014; 16 (5): 470. https://doi.org/10.1186/s13075-014-0470-8.
49. Shi Q., Liu H., Tang D., et al. Bioactuators based on stimulus-responsive hydrogels and their emerging biomedical applications. NPG Asia Materials. 2019; 11: 64. https://doi.org/10.1038/s41427-019-0165-3.
50. Saboowala H. (Ed.) What is neural encoding and neural decoding? Neural decoding to decipher brain activity to treat paralysis, rheumatoid arthritis and epilepsy. 2019.
51. Yaprak M. The axon reflex. Neuroanatomy. 2008; 7: 17–9.
52. Barnes P. Asthma as an axon reflex. Lancet. 1986; 1 (8475): 242–5. https://doi.org/10.1016/s0140-6736(86)90777-4.
53. Houghton B.L., Meendering J.R., Wong B.J., Minson C.T. Nitric oxide and noradrenaline contribute to the temperature threshold of the axon reflex response to gradual local heating in human skin. J Physiol. 2006; 572 (Pt 3): 811–20. https://doi.org/10.1113/jphysiol.2005.104067.
54. Charkoudian N., Wallin B.G. Sympathetic neural activity to the cardiovascular system: integrator of systemic physiology and interindividual characteristics. Compr Physiol. 2014; 4 (2): 825–50. https://doi.org/10.1002/cphy.c130038.
55. Dampney R.A. Central neural control of the cardiovascular system: current perspectives. Adv Physiol Educ. 2016; 40 (3): 283–96. https://doi.org/10.1152/advan.00027.2016.
56. Mayer E.A. Gut feelings: the emerging biology of gut-brain communication. Nat Rev Neurosci. 2011; 12 (8): 453–66. https://doi.org/10.1038/nrn3071.
57. Travagli R.A., Hermann G.E., Browning K.N., Rogers R.C. Brainstem circuits regulating gastric function. Annu Rev Physiol. 2006; 68: 279–305. https://doi.org/10.1146/annurev.physiol.68.040504.094635.
58. Krohn K., Uibo R., Aavik E., et al. Identification by molecular cloning of an autoantigen associated with Addison's disease as steroid 17 alpha-hydroxylase. Lancet. 1992; 339 (8796): 770–3. https://doi.org/10.1016/0140-6736(92)91894-e.
59. Hosoi T., Okuma Y., Matsuda T., Nomura Y. Novel pathway for LPS-induced afferent vagus nerve activation: possible role of nodose ganglion. Auton Neurosci. 2005; 120 (1-2): 104–7. https://doi.org/10.1016/j.autneu.2004.11.012.
60. Xu Z.Z., Kim Y.H., Bang S., et al. Inhibition of mechanical allodynia in neuropathic pain by TLR5-mediated A-fiber blockade. Nat Med. 2015; 21 (11): 1326–31. https://doi.org/10.1038/nm.3978.
61. Foley J.O., DuBois F.S. Quantitative studies of the vagus nerve in the cat: I. The ratio of sensory to motor fibers. J Compar Neurol. 1937; 67 (1): 49–67. https://doi.org/10.1002/cne.900670104.
62. Ethier C., Oby E.R., Bauman M.J., Miller L.E. Restoration of grasp following paralysis through brain-controlled stimulation of muscles. Nature. 2012; 485 (7398): 368–71. https://doi.org/10.1038/nature10987.
63. Park C.K., Xu Z.Z., Berta T., et al. Extracellular microRNAs activate nociceptor neurons to elicit pain via TLR7 and TRPA1. Neuron. 2014; 82 (1): 47–54. https://doi.org/10.1016/j.neuron.2014.02.011.
64. Steinberg B.E., Silverman H.A., Robbiati S., et al. Cytokine-specific neurograms in the sensory vagus nerve. Bioelectron Med. 2016; 3: 7–17.
65. Kawashima K., Fujii T., Moriwaki Y., Misawa H. Critical roles of acetylcholine and the muscarinic and nicotinic acetylcholine receptors in the regulation of immune function. Life Sci. 2012; 91 (21–22): 1027–32. https://doi.org/10.1016/j.lfs.2012.05.006.
66. Kawashima K., Fujii T., Moriwaki Y., et al. Non-neuronal cholinergic system in regulation of immune function with a focus on α7nAChRs. Int Immunopharmacol. 2015; 29 (1): 127–34. https://doi.org/10.1016/j.intimp.2015.04.015.
67. Talbot S., Foster S.L., Woolf C.J. Neuroimmunity: physiology and pathology. Annu Rev Immunol. 2016; 34: 421–47. https://doi.org/10.1146/annurev-immunol-041015-055340.
68. Pinho-Ribeiro F.A., Verri W.A. Jr., Chiu I.M. Nociceptor sensory neuron-immune interactions in pain and inflammation. Trends Immunol. 2017; 38 (1): 5–19. https://doi.org/10.1016/j.it.2016.10.001.
69. Lai N.Y., Mills K., Chiu I.M. Sensory neuron regulation of gastrointestinal inflammation and bacterial host defence. J Intern Med. 2017; 282 (1): 5–23. https://doi.org/10.1111/joim.12591.
70. Julius D. TRP channels and pain. Annu Rev Cell Dev Biol. 2013; 29: 355–84. https://doi.org/10.1146/annurev-cellbio-101011-155833.
71. Bollella P., Fusco G., Tortolini C., et al. Beyond graphene: electrochemical sensors and biosensors for biomarkers detection. Biosens Bioelectron. 2017; 89 (Pt 1): 152–66. https://doi.org/10.1016/j.bios.2016.03.068.
About the Authors
M. DhallIndia
Manish Dhall, M. Pharm., PhD
R. Tushir
India
Renu Tushir, M. Pharm.
P. Sharma
India
Prerna Sharma, Assoc. Prof.
N. Rani
India
Nidhi Rani, M. Pharm., PhD, Assoc. Prof
T. Singh
India
Thakur Gurjeet Singh, Prof.
Review
For citations:
Dhall M., Tushir R., Sharma P., Rani N., Singh T. Bioelectronic medicine: a new frontier for autonomic nervous system disorders. Epilepsy and paroxysmal conditions. 2025;17(3):297-307. https://doi.org/10.17749/2077-8333/epi.par.con.2025.231
JATS XML

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.










































